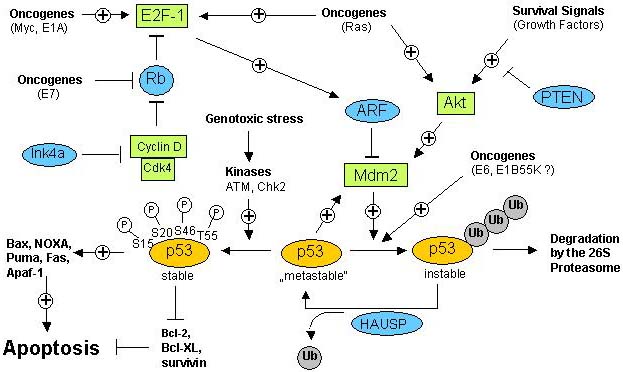
Figure 8
The p53 network ñ survival and cell death regulation.
In a normal growing viable cell, the p53 protein is in a metastable state, i.e. p53 is susceptible to targeted ubiquitination and subsequent proteasomal degradation. Mdm2 directly interacts with p53 and thereby catalyzes ubiquitination of p53. Ubiquitination of p53 can be reversed by the action of the deubiquitinating enzyme HAUSP which thereby can rescue p53 from degradation (see also paragraph 1.2.3.2). p53 is stabilized in response to genotoxic stress such as DNA damage which leads to the phosphorylation of p53 at several specific serine and threonine residues. Stabilized and activated p53 can translocate into the nucleus where it activates the transcription of proapoptotic genes and suppresses the transcription of antiapoptotic genes what under certain conditions can result in the induction of apoptosis. p53-mediated apoptosis signalling is dependent on the interplay of many regulatory factors, including protooncogenes as well as tumor-suppressors. Mdm2 activity is positively regulated by the action of the Akt kinase: when Mdm2 is phophorylated by Akt, Mdm2 is able to translocate from the cytosol to the nucleus where it unfolds its inhibitory effect on p53. Akt kinase, on the other hand, is activated in response to survival signals coming from growth factor receptors. This is therefore an instructive example for the negative regulation of proapoptotic, p53-mediated signals by survival signalling. Whereas Akt kinase positively regulates Mdm2 activity, Mdm2-mediated suppression of p53 is blocked by the a ction of the ARF tumor suppressor. By binding to Mdm2, ARF prevents the interaction between Mdm2 and p53 and therefore stabilizes and activates p53. ARF expression is dependent on the transcription factor E2F-1 which is regulated by the retinoblastoma (Rb) tumor-suppressor and by the action of oncogenes. As an example, mitogenic signals lead to the activation of oncogenes such as c-myc and ras which among others activate E2F-1, resulting in increased ARF activity, stabilization of p53 and induction of apoptosis. Therefore, increased mitogenic signalling or inappropriate oncogenic activity not necessarily causes excessive proliferation but in cells with intact p53 signalling pathways can act as apoptosis inducers.
The p53 network ñ survival and cell death regulation.
In a normal growing viable cell, the p53 protein is in a metastable state, i.e. p53 is susceptible to targeted ubiquitination and subsequent proteasomal degradation. Mdm2 directly interacts with p53 and thereby catalyzes ubiquitination of p53. Ubiquitination of p53 can be reversed by the action of the deubiquitinating enzyme HAUSP which thereby can rescue p53 from degradation (see also paragraph 1.2.3.2). p53 is stabilized in response to genotoxic stress such as DNA damage which leads to the phosphorylation of p53 at several specific serine and threonine residues. Stabilized and activated p53 can translocate into the nucleus where it activates the transcription of proapoptotic genes and suppresses the transcription of antiapoptotic genes what under certain conditions can result in the induction of apoptosis. p53-mediated apoptosis signalling is dependent on the interplay of many regulatory factors, including protooncogenes as well as tumor-suppressors. Mdm2 activity is positively regulated by the action of the Akt kinase: when Mdm2 is phophorylated by Akt, Mdm2 is able to translocate from the cytosol to the nucleus where it unfolds its inhibitory effect on p53. Akt kinase, on the other hand, is activated in response to survival signals coming from growth factor receptors. This is therefore an instructive example for the negative regulation of proapoptotic, p53-mediated signals by survival signalling. Whereas Akt kinase positively regulates Mdm2 activity, Mdm2-mediated suppression of p53 is blocked by the a ction of the ARF tumor suppressor. By binding to Mdm2, ARF prevents the interaction between Mdm2 and p53 and therefore stabilizes and activates p53. ARF expression is dependent on the transcription factor E2F-1 which is regulated by the retinoblastoma (Rb) tumor-suppressor and by the action of oncogenes. As an example, mitogenic signals lead to the activation of oncogenes such as c-myc and ras which among others activate E2F-1, resulting in increased ARF activity, stabilization of p53 and induction of apoptosis. Therefore, increased mitogenic signalling or inappropriate oncogenic activity not necessarily causes excessive proliferation but in cells with intact p53 signalling pathways can act as apoptosis inducers.